Dystrogen Therapeutics
Private Company
Total funding raised: $4M
Overview
Dystrogen Therapeutics is developing a first-in-class chimeric cell therapy platform with broad applications in genetic and degenerative disorders. Its core technology involves fusing donor and recipient cells to create 'Dystrophin Expressing Chimeric (DEC)' cells that evade immune rejection, eliminating the need for immunosuppression. The company is initially focused on Duchenne Muscular Dystrophy, with a Phase 1 trial underway, and has preclinical programs targeting sarcopenia, age-related muscle loss, and neurodegenerative diseases. Dystrogen's approach represents a potentially paradigm-shifting alternative to gene therapy and other advanced modalities.
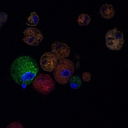
Technology Platform
A chimeric cell engineering platform that fuses donor and recipient cells to create therapeutic cells that produce missing proteins (e.g., dystrophin) while evading immune rejection, eliminating the need for immunosuppression.
Funding History
2Opportunities
Risk Factors
Competitive Landscape
Dystrogen competes with gene therapy leaders like Sarepta and Pfizer in DMD, as well as exon-skipping and stop-codon readthrough approaches. Its main differentiation is offering a universal, immunosuppression-free therapy that delivers full-length dystrophin without genetic manipulation or viral vectors, potentially offering a superior safety and efficacy profile.